Mayo Clinic Discovery Square
Labworks International recently completed manufacturing a pharmaceutical grade cleanroom for the Mayo Clinic at their Facility for Advance Regenerative Biomanufacturing.
Learn More
Available in multiple custom configurations, the LABTECH* ECO-Series offers the world's most eco-friendly environmental chambers with precision-controlled temperature and humidity.
Labworks Stability Chambers are engineered with flexibility to meet 2-8C, 25C/60, 30C/70, 40C/75 and freezer applications. The Labtech™ advanced refrigeration system offers precise control while keeping all of the refrigeration equipment out of the stability chamber. In large applications a CFD model of the proposed stability chamber is constructed, which ensures the final construction performs as expected.

Labworks environmental room solutions include freezer spaces ranging from small walk-in rooms to very large warehouse-style spaces. Labworks can deliver environmental room freezers with temperature performance as low as -80C. For critical spaces requiring precise control, Labworks has developed our constant temperature freezer design which can complete defrost cycles while maintaining design temperatures for the freezer room.

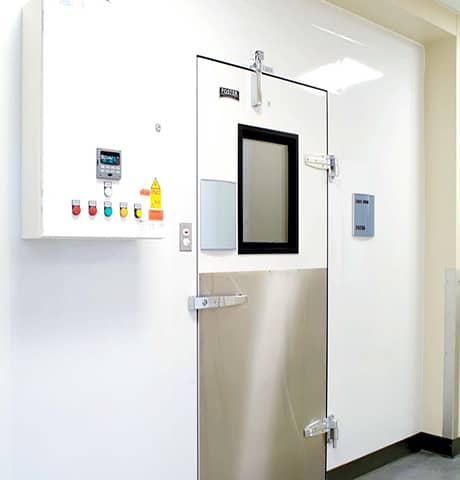
Validated clean rooms are our specialty. Stringent requirements on cleanliness combined with strict temperature performance make proper engineering a critical factor with GMP grade clean rooms. Our designers ensure temperature and particle performance are compliant with the regulations for the designated grade of clean room. A variety of panel surface coatings are available to suit the process requirements of the area.

Labworks specializes in critical clean room applications. Our clean room experience includes applications utilized for research, laboratories, healthcare and forensics. Every installation is professionally engineered to ensure optimum performance; clean room users are carefully surveyed to ensure the design incorporates all of their needs. Special attention is paid to serviceability of the clean room space, ensuring systems can be properly maintained over the life of the equipment.

Labworks International Modular Manufacturing Facility provides refabricated Environmental Rooms, Test Chambers, Air Locks, Classifies Cold/ Warm Rooms, and Pass-Throughs. The units are designed, assembled and tested in Labworks’ facility prior to shipment to clients across North America.

Labworks takes Cold Chain Storage design to the next level; utilizing CFD computer models to ensure what we build works. Our solutions are fully redundant, efficient, and can be customized to your space and temperature needs. The Labworks cold chain facility design starts with a comprehensive needs assessment to feed the design model, ensuring uniform performance across the entire space.

Labworks has completed some of the largest mortuary facilities in North America. Labworks offers complete turn-key solutions for mortuary rooms. Design/build installations include room construction, cooler panels, refrigeration equipment, rack assemblies, lift solutions, autopsy tables, and all other equipment required to fit-out the room.

Labworks Scientific Grade Reach-In chambers are ideal for storage and testing of products requiring precision temperature and humidity control. Designed to comply with FDA (21 CFR part 11) and Health Canada standards, these units can be part of a validated (ICH) product storage system for storage of pharmaceutical of other temperature sensitive products, or as part of a process requiring conditioning of material under strict conditions.
Labworks International Inc. are Professional Engineers, Constructors and Manufacturers of Environmental Room solutions.
We provide turn-key solutions throughout North America, and are experts in designing and constructing precision controlled environments for the safe and reliable storage of temperature and/or humidity sensitive items.
We focus on the life sciences industries such as healthcare, post-secondary institutions, pharmaceutical, government research facilities and private industry.
UHN Toronto's DriverLab is the most advanced driving simulator in Canada and unique in the world. Labworks was honored to have engineered and installed the HVAC and controls required for this unique simulator. Specialized HVAC equipment was required to be installed in a challenging and congested existing mechanical space as part of the project which was achieved successfully.
Precise temperature control in the simulator was achieved through sophisticated controls and innovative supply and return air designs to allow for a wide range of temperature performance set points. Traditional HVAC ducting and controls materials and equipment could not be utilized as the simulator was required to be moved to different positions throughout the facility. ‘Quick' change and flexible connections were required on the ducting along with the controls for the simulator. All of the specialized components came together without exception to achieve yet another successful and unique project by Labworks International.
Through Labworks' Revit software training development program, we develop intelligent Building Information that will improve communication and collaboration with our customers and contractors. This will, in turn, reduce the turn-around time for project deliverables.
We can streamline the design process with the use of a central 3D model, where changes made in one view will simultaneously update across all views and on the printable sheets.
This improved operational agility will enhance Labworks International's business competitiveness in Canadian and international markets.
Labworks is an international leading supplier of specialty environments. We are a leading provider in Canada and the US and have continued requirements for high-quality candidates who want to grow their careers in a fast-paced and exciting environment. Take a look at the positions below, if interested please send over your resume to admin@labworksinternational.com

Labworks International maintains strict quality control measures throughout the manufacturing process. We utilize high-quality materials, advanced technologies, and rigorous testing to ensure that our environmental rooms meet industry standards and deliver reliable performance.
Absolutely! Labworks International specializes in providing customized solutions for unique environmental room requirements. Our team of experts will work closely with you to understand your specific needs and design a tailored environmental room that meets your exact specifications.
The manufacturing and installation time for an environmental room can vary depending on factors such as size, complexity, and customization requirements. Labworks International will provide you with a detailed project timeline and keep you informed throughout the process.
Yes, Labworks International offers comprehensive maintenance and support services for our environmental rooms. We provide regular maintenance checks, calibration services, and prompt assistance for any issues or inquiries you may have.